Diagnosis of coeliac disease is based on four pillars. It is essential that patients follow a gluten-containing diet both before and during the diagnostic process.
The four pillars underpinning a diagnosis of coeliac disease, are:
1. Clinical history
Clinical history includes both personal and family medical history in addition to a dietary history that specifically focuses on foods that contain gluten. Clinical symptoms are also monitored in this step, for example bowel habits, weight loss, abdominal bloating, abdominal pain, nausea and growth disorders in children. Extraintestinal symptoms such as fatigue, bone/joint pain and headaches/migraine are also important factors to consider here.
2. Serological tests
Serological testing for coeliac disease involves the identification of immunoglobulin A tissue transglutaminase (IgA tTG) antibodies and specific endomysial antibodies (EMA). Total IgA should be determined in order to rule out IgA deficiency and reduce the risk of a false negative result. In patients with known IgA deficiency, immunoglobulin G (IgG) EMA, IgG deaminated gliadin peptide (DGP) or IgG tTG may be used in order to support the diagnosis [1,2]. It is essential that patients consume a gluten containing diet prior to and during diagnostic investigation as results may normalise on a gluten-free diet.
HLA typing
Human leukocyte antigen (HLA) DQ2/DQ8 testing may be considered within the diagnostic work-up for patients within specialist settings. The diagnostic value of HLA genotyping within this patient group, revolves around it’s high negative predictive value – a negative result indicating that a patient is highly unlikely to have coeliac disease (less than 1% of patients with coeliac disease do not carry these alleles [3]). However, the positive predictive value of value of HLA genotyping is very low, since a up to 40% of the general population also carry genes encoding HLA DQ2/DQ8 [4]. HLA testing may therefore be useful for patients where the diagnosis is equivocal, or for those who have already embarked on a gluten free diet and choose not to have a gluten challenge.
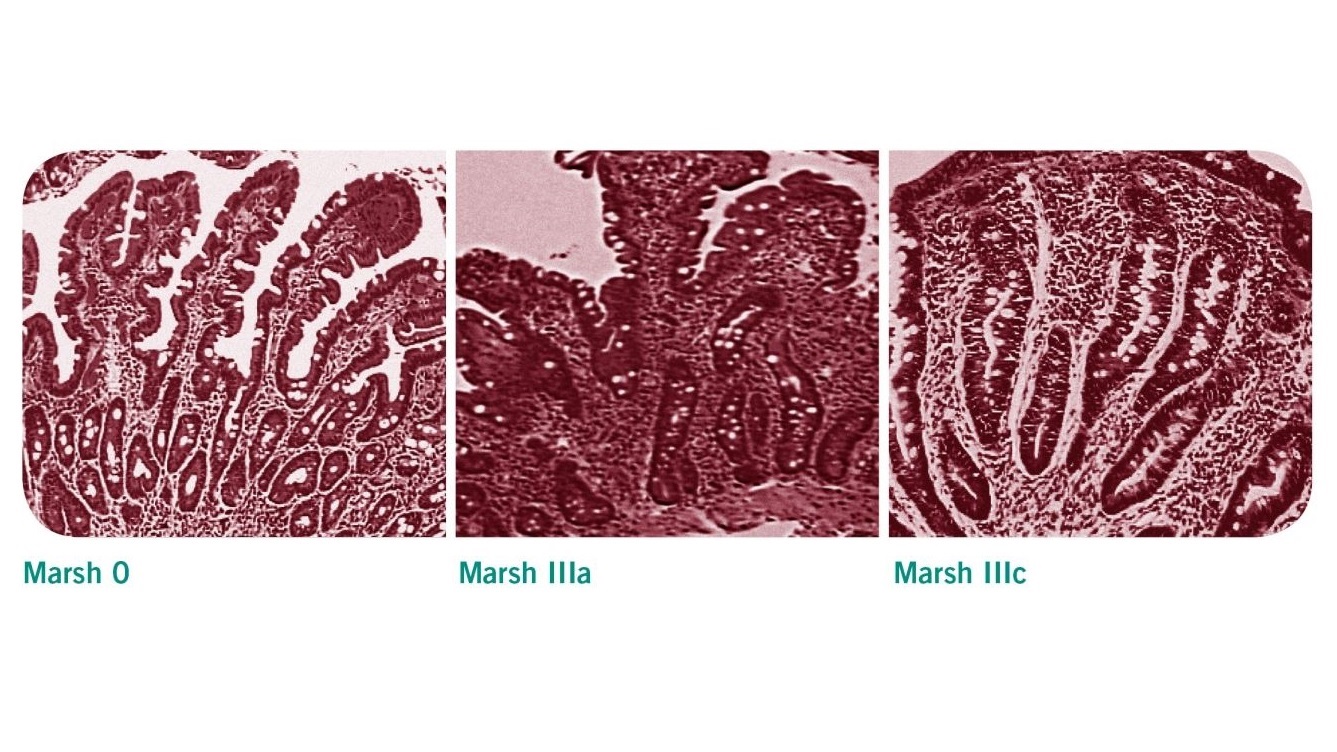
3. Histological tests
Patients with positive serological test results should be referred to a gastrointestinal specialist for endoscopic intestinal biopsy to confirm or exclude coeliac disease [2]. Villous atrophy may be patchy in coeliac disease, therefore a minimum of four biopsy specimens should be obtained, including a duodenal bulb biopsy [1]. The histological changes observed in these samples are classified according to the Marsh classification. Evidence of villous atrophy and increased intraepithelial lymphocytes are typical features of a coeliac-positive histology (Marsh 3a-c).
| Marsh classification | histological findings |
|---|---|
| Stage 0 | Normal duodenal mucosa |
| Stage 1 | Increased intraepithelial lymphocytes (IELs) >25 IELs/100 enterocytes (non-specific finding) |
| Stage 2 | Stage 1 plus crypt hyperplasia (non-specific finding) |
| Stage 3a | Increased IELs, crypt hyperplasia, and partial villous atrophy |
| Stage 3b | Increased IELs, crypt hyperplasia, and subtotal villous atrophy |
| Stage 3c | Increased IELs, crypt hyperplasia, and total villous atrophy |
4. Response to a gluten-free diet
The diagnosis of coeliac disease is considered to be confirmed if symptoms improve and repeat serological testing indicates that the antibodies are responding to the gluten-free diet. Current NICE guidelines on the Recognition, Assessment and Management of Coeliac Disease [2] recommend that patients with persistently high serological titres or persistent symptoms after 12 months (where the possibility of continued gluten exposure has been excluded) should be considered for repeat intestinal biopsy and review by a specialist gastroenterology team.
Regular follow-up examinations
Guidelines from the Primary Care Society for Gastroenterology (PCSG) [8] recommend that newly diagnosed patients should be reviewed after 3-6 months and annually thereafter in order to monitor compliance with and response to treatment, and evidence of disease complications. Patients should be offered access to specialist dietetic and nutritional advice as part of this review, during which symptoms may be reviewed alongside dietary adherence, nutritional adequacy, and collection of anthropometrical data [2]. Repeat serological testing may be used in conjunction with dietetic review in order to assess adherence. Follow-up biopsies are not routinely used in the review of patients with coeliac disease but may be useful for patients whose condition does not respond to a gluten free diet [1].
Reference
- Ludvigsson JF, Bai JC, Biagi F et al. Diagnosis and management of adult coeliac disease : guidelines from the British Society of Gastroenterology. Gut 2014; 63(8):1210-28
- NICE Guideline NG20: Recognition, Assessment & Management of Coeliac Disease. National Institute of Clinical Excellence 2015.
- Leffler D, Schuppan D, Pallav K, et al. Kinetics of the histological, serological and symptomatic responses to gluten challenge in adults with coeliac disease. Gut 2013; 62: 996–1004
- Polvi A, Arranz E, Fernandez-Arquero M et al. HLA-DQ2-negative celiac disease in Finland and Spain. Hum Immunol 1998;59:169-75
- Abadie V, Sollid LM, Barreiro LB et al. Integration of genetic and immunological insights into a model of celiac disease pathogenesis. Annu Rev Immunol 2011; 29:493-525
- Oberhuber G, Granditsch G, Vogelsang H. The histopathology of coeliac disease: time for a standardized report scheme for pathologists. Eur J Gastroenterol Hepatol 1999; 11:1185-94
- Husby S, Koletzko S, Korponay-Szabó IR et al. European Society for Pediatric Gastroenterology, Hepatology, and Nutrition guidelines for the diagnosis of coeliac disease. J Pediatr Gastroenterol Nutr 2012; 54: 136-160
- Murch S, Jenkins H, Auth M et al. Joint BSPGHAN and Coeliac UK guidelines for the diagnosis and management of coeliac disease in children. Arch Dis Child 2013 98: 806-811.
- The Management of Adults with Coeliac Disease in Primary Care. Primary Care Society for Gastroenterology 2006.
